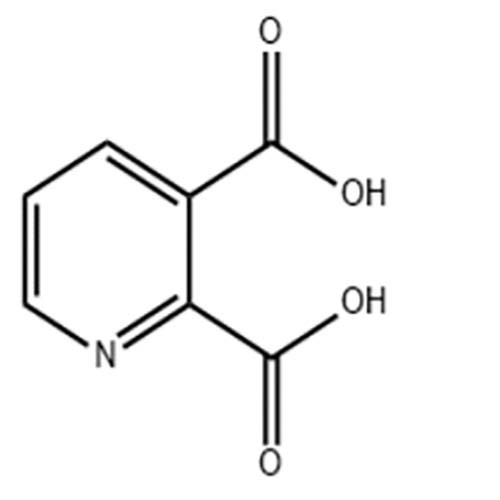
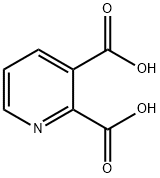
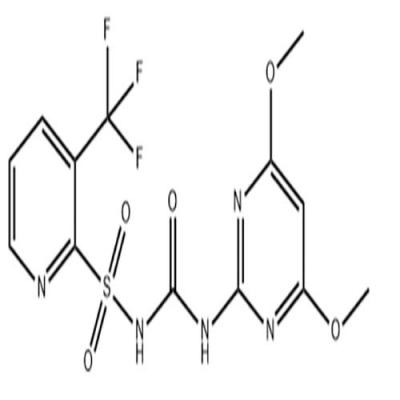
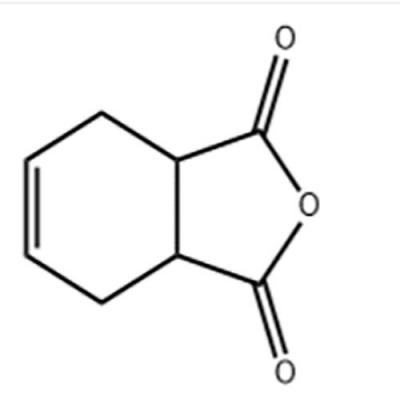
Welcome: Wuhan Yihe Technology Co., Ltd
![af]() Afrikaans
Afrikaans![sq]() Albanian
Albanian![am]() Amharic
Amharic![ar]() Arabic
Arabic![fr]() French
French![es]() Spanish
Spanish![ru]() Russian
Russian![de]() German
German![hy]() Armenian
Armenian![it]() Italian
Italian![ja]() Japanese
Japanese![ko]() Korean
Korean![pt]() Portuguese
Portuguese![hi]() Hindi
Hindi![az]() Azerbaijani
Azerbaijani![ro]() Romanian
Romanian![pl]() Polish
Polish![th]() Thai
Thai![el]() Greek
Greek![eu]() Basque
Basque![en]() English
English![zh-CN]() Chinese (Simplified)
Chinese (Simplified)![zh-TW]() Chinese (Traditional)
Chinese (Traditional)![be]() Belarusian
Belarusian![bn]() Bengali
Bengali![bs]() Bosnian
Bosnian![bg]() Bulgarian
Bulgarian![ca]() Catalan
Catalan![ceb]() Cebuano
Cebuano![ny]() Chichewa
Chichewa![co]() Corsican
Corsican![hr]() Croatian
Croatian![cs]() Czech
Czech![da]() Danish
Danish![nl]() Dutch
Dutch![eo]() Esperanto
Esperanto![et]() Estonian
Estonian![tl]() Filipino
Filipino![fi]() Finnish
Finnish![fy]() Frisian
Frisian![gl]() Galician
Galician![ka]() Georgian
Georgian![gu]() Gujarati
Gujarati![ht]() Haitian Creole
Haitian Creole![ha]() Hausa
Hausa![haw]() Hawaiian
Hawaiian![iw]() Hebrew
Hebrew![hmn]() Hmong
Hmong![hu]() Hungarian
Hungarian![is]() Icelandic
Icelandic![ig]() Igbo
Igbo![id]() Indonesian
Indonesian![ga]() Irish
Irish![jw]() Javanese
Javanese![kn]() Kannada
Kannada![kk]() Kazakh
Kazakh![km]() Khmer
Khmer![ku]() Kurdish (Kurmanji)
Kurdish (Kurmanji)![ky]() Kyrgyz
Kyrgyz![lo]() Lao
Lao![la]() Latin
Latin![lv]() Latvian
Latvian![lt]() Lithuanian
Lithuanian![lb]() Luxembourgish
Luxembourgish![mk]() Macedonian
Macedonian![mg]() Malagasy
Malagasy![ms]() Malay
Malay![ml]() Malayalam
Malayalam![mt]() Maltese
Maltese![mi]() Maori
Maori![mr]() Marathi
Marathi![mn]() Mongolian
Mongolian![my]() Myanmar (Burmese)
Myanmar (Burmese)![ne]() Nepali
Nepali![no]() Norwegian
Norwegian![ps]() Pashto
Pashto![fa]() Persian
Persian![pa]() Punjabi
Punjabi![sm]() Samoan
Samoan![gd]() Scottish Gaelic
Scottish Gaelic![sr]() Serbian
Serbian![st]() Sesotho
Sesotho![sn]() Shona
Shona![sd]() Sindhi
Sindhi![si]() Sinhala
Sinhala![sk]() Slovak
Slovak![sl]() Slovenian
Slovenian![so]() Somali
Somali![su]() Sudanese
Sudanese![sw]() Swahili
Swahili![sv]() Swedish
Swedish![tg]() Tajik
Tajik![ta]() Tamil
Tamil![te]() Telugu
Telugu![tr]() Turkish
Turkish![uk]() Ukrainian
Ukrainian![ur]() Urdu
Urdu![uz]() Uzbek
Uzbek![vi]() Vietnamese
Vietnamese![cy]() Welsh
Welsh![xh]() Xhosa
Xhosa![yi]() Yiddish
Yiddish![yo]() Yoruba
Yoruba![zu]() Zulu
Zulu